Three potential Covid-19 vaccines have reported they may be about 90 per cent effective.
The vaccine being developed by the University of Oxford is up to 90 per cent effective in preventing coronavirus, according to early data from phase three of the clinical trial.
Recent data from the Pfizer/BioNTech and Moderna vaccine trials suggests their candidates are 95 per cent and 94.5 per cent effective respectively.
But how does the Oxford vaccine compare with these?
What has been announced?
Oxford data indicates the vaccine has 62 per cent efficacy when one full dose is given followed by another full dose.
But when people were given a half dose followed by a full dose at least a month later, its efficacy rose to 90 per cent.
The combined analysis from both dosing regimes resulted in an average efficacy of 70.4 per cent.
Don’t vaccines take a long time to produce?
In the past it has taken years, sometimes decades, to produce a vaccine.
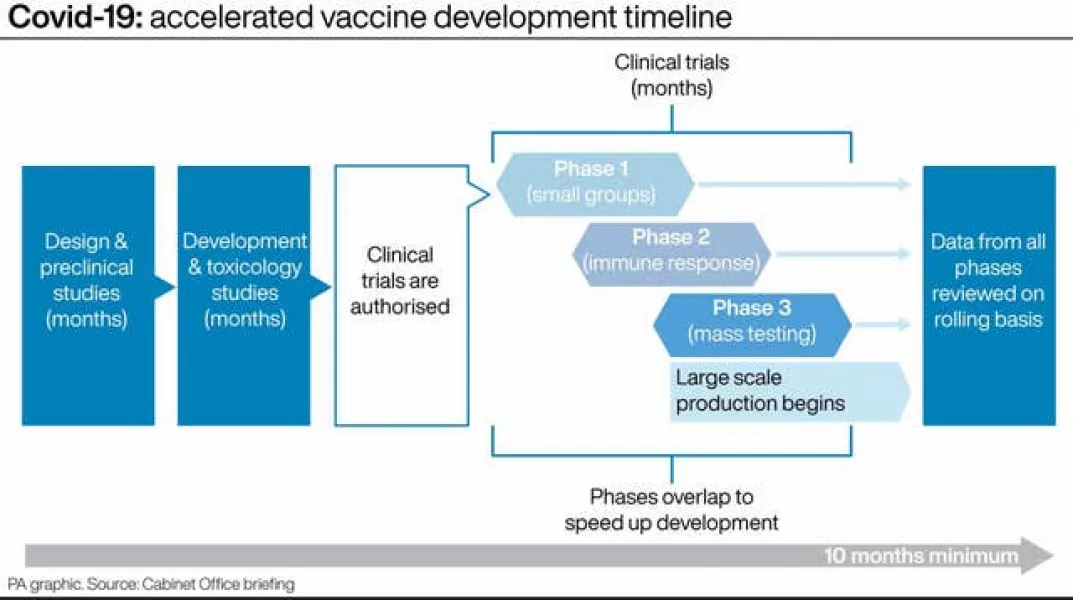
Traditionally, vaccine development includes various processes, including design and development stages followed by clinical trials – which in themselves need approval before they even begin.
But in the trials for a Covid vaccine, things look slightly different. A process which usually takes years has been condensed to months.
While the early design and development stages look similar, the clinical trial phases overlap, instead of taking place sequentially.
And pharmaceutical firms have begun manufacturing before final approval has been granted – taking on the risk that they may be forced to scrap their work.
The new way of working means that regulators around the world can start to look at scientific data earlier than they traditionally would do.
Aren’t there two other vaccines?
Yes, recent data from the Pfizer/BioNTech and Moderna vaccine trials suggests their candidates are 95 per cent and 94.5 per cent effective respectively.
Pfizer, with its partner BioNTech, is expected to receive US approval for its vaccine within days, while phase three trials of the Moderna vaccine continue.
Which jab is best?
Data from the Moderna trials indicate 94.5 per cent effectiveness, but this number could change as the trials are ongoing.
Early results from Pfizer found it is 95 per cent effective within 28 days of the first dose.
It was also 94 per cent effective in those aged 65 and over – those most at risk from Covid-19.
But Oxford researchers say it is difficult to make direct comparisons because it is not yet known exactly what everyone is measuring in the trials.
What do they cost?
Researchers suggest the Oxford vaccine could be relatively cheap to produce, with some reports indicating it could be about £3 (€3.38) per dose.
AstraZeneca said it will not sell it for a profit, so it can be available to all countries.
Pfizer/BioNTech is also making its vaccine available not-for-profit.
According to reports, the Moderna vaccine could cost about $38 US dollars (€32) per dose, and the Pfizer candidate could cost around $20 (€17).
How do we know the vaccines are safe?
Full safety data has not been made available yet, but researchers report their trials do not suggest any significant safety concerns.
Despite the speed of which the Covid-19 vaccines have been developed, all safety measures have been observed.
Who gets which vaccine?
As things stand, the vaccines will be rolled out as and when they become available.
No announcement has been made on whether one might be given priority over another as they become ready on a mass scale.
A taskforce has been established to determine the most effective way to administer the vaccine, with members meeting this week to plan potential roll-out methods.
Speaking last week, the chairman of the taskforce, Professor Brian MacCraith said a strategy to vaccinate the Irish population will be dealt with “urgently and comprehensively”.
Prof MacCraith added the taskforce will release information on the roll out of vaccines when it is “available and when it’s accurate”.
It is not expected that people will be able to choose which vaccine they receive.
When will we get the vaccines?
No vaccine has been approved for use yet, but the jab is likely to be rolled out in the EU from December, with the bulk of vaccination in the new year.
Are other vaccines on the way?
Around the world, scientists continue to work on developing other vaccines, with experts agreeing that more than one will be needed.
Other vaccines are also being developed by Novavax, Janssen and Valneva.
Vaccines being developed in China and Russia have also shown positive results in early trial findings
When might we return to normal life?
While it is not known exactly how long it might take for the population to be vaccinated, Dublin-born vaccinologist Professor Adrian Hill, who is head of the Jenner Institute in Oxford, has said that “maybe by next May or June we could be back to normal”.
However, Prof Hill added that until there was widespread vaccination, people would have to continue adhering to public health guidelines, including wearing face coverings, socially distancing and washing their hands.